Provide lookups for medical devices from the Australian ARTG and US FDA GUDID databases
Export structured data for medical devices from the Australian ARTG (~62,500 devices) and US FDA GUDID (~4M devices), with cross-registry matching to build an AU-US device lookup table.
What This Does
- Australia (ARTG): Scrapes the TGA website for device IDs, downloads PDF summaries, parses them into structured JSON
- USA (FDA): Downloads GUDID bulk data and openFDA classification, parses pipe-delimited files into structured JSON
- Matching:Links AU and US devices using GMDN codes, manufacturer names, device descriptions, and risk classification
Scoring Algorithm
Each ARTG device is scored against candidate FDA devices. Candidates are found via the GMDN inverted index (only devices sharing at least one GMDN code are considered).
Score Components
| Signal | Weight | Range | Method |
|---|---|---|---|
| GMDN code match | 0.50 | 0 or 0.50 | Any overlap in GMDN codes between ARTG products and FDA device |
| Manufacturer name | 0.30 | 0.0-0.30 | rapidfuzz.fuzz.token_sort_ratio + partial_ratio, best score across all ARTG manufacturers vs FDA company |
| Device description | 0.15 | 0.0-0.15 | rapidfuzz.fuzz.token_set_ratio on ARTG intended purpose vs FDA device description |
| Risk class compatibility | 0.05 | 0 or 0.05 | ARTG class maps to compatible FDA class(es) |
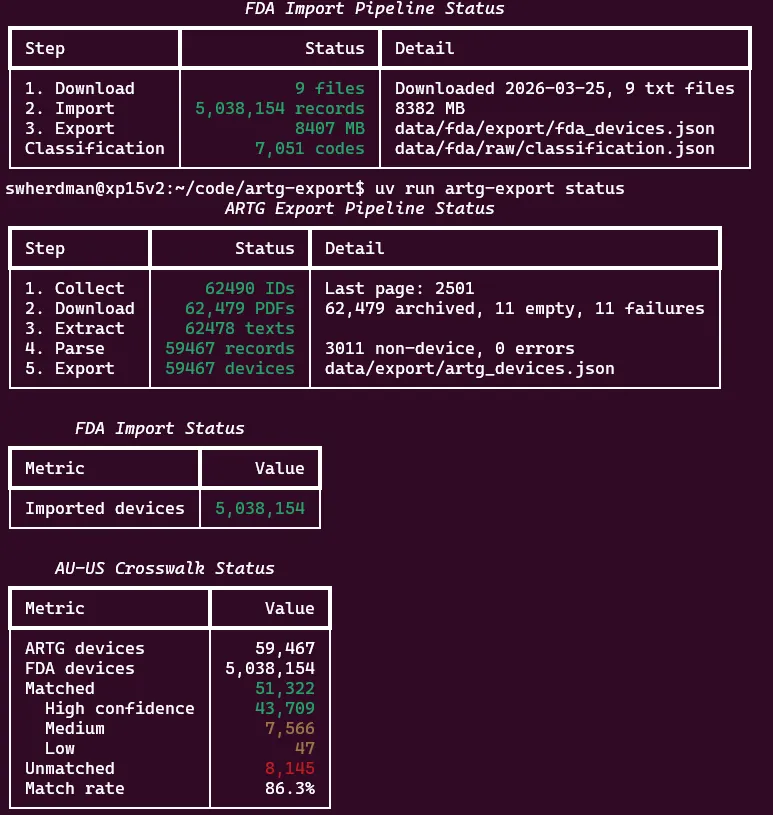
$ uv run artg-export match-status
AU-US Device Crosswalk Status
┏━━━━━━━━━━━━━━━━━━━━━┳━━━━━━━━━━━━━━━━━━━━━┓
┃ Metric ┃ Value ┃
┡━━━━━━━━━━━━━━━━━━━━━╇━━━━━━━━━━━━━━━━━━━━━┩
│ Generated at │ 2026-03-26T00:42:36 │
│ ARTG devices │ 59,467 │
│ FDA devices │ 5,038,154 │
│ │ │
│ Matched (any) │ 51,322 │
│ High confidence │ 43,709 │
│ Medium confidence │ 7,566 │
│ Low confidence │ 47 │
│ Unmatched │ 8,145 │
│ │ │
│ Match rate │ 86.3% │
└─────────────────────┴─────────────────────┘